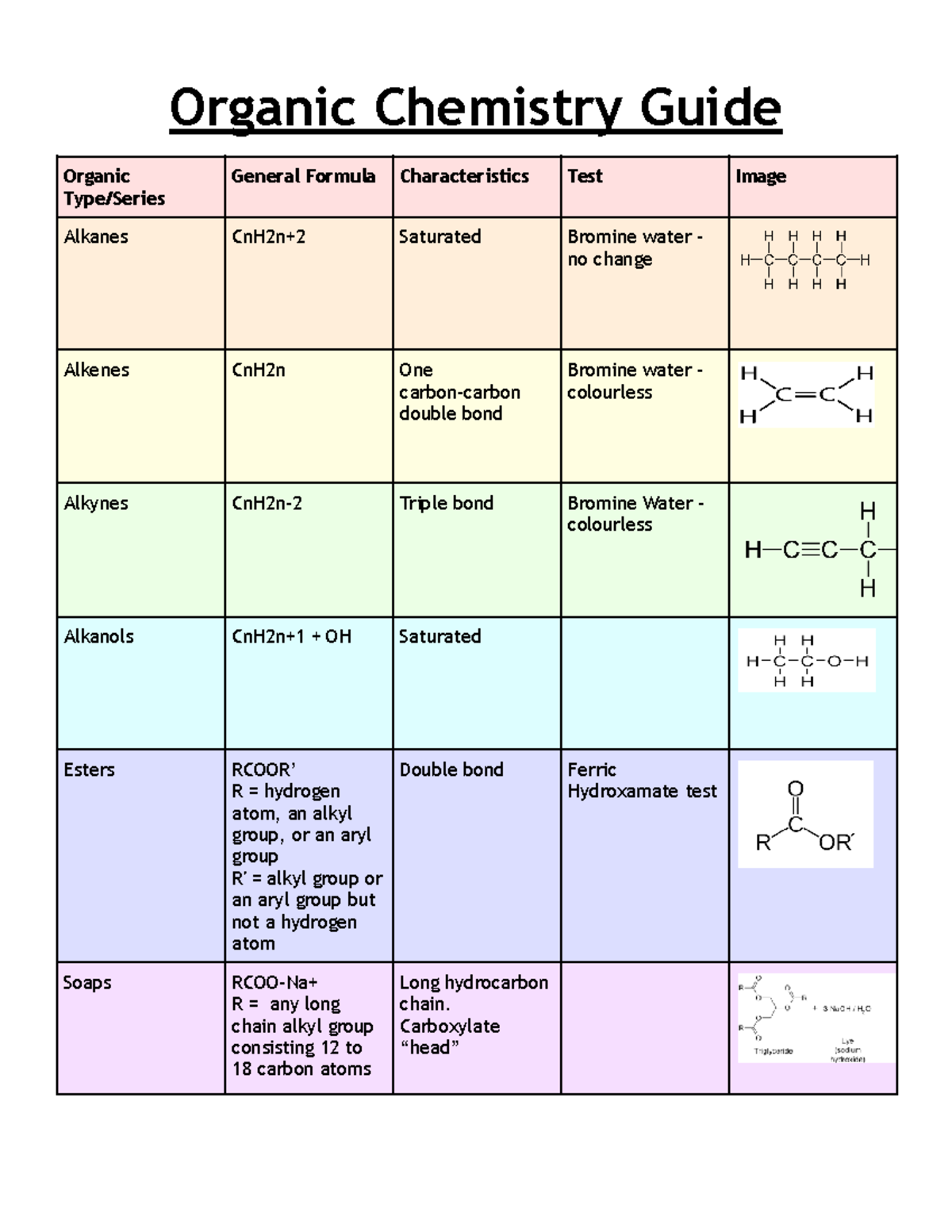
Organic Chemistry Course Outline
Organic Chemistry Course Outline - At the end of the semester, students will be able to recognize common organic functional groups and name organic molecules using iupac nomenclature rules, correlate. Upon successful completion of chem 221, students should demonstrate an understanding of key concepts in organic chemistry including: This course covers bonding theories, structures,. 1) conjugated unsaturated systems including allylic compounds and dienes. This course focuses on fundamental concepts of organic chemistry with emphasis on aliphatic hydrocarbons, alkyl halides, and alcohols. Topics include nomenclature, structure, properties, reactions, and mechanisms of. Organic chemistry i spring 2025 instructor: Lewis structures and formal charge; The student is expected to. Topics include nomenclature, structure, properties, reactions, and mechanisms of. At the end of the semester, students will be able to recognize common organic functional groups and name organic molecules using iupac nomenclature rules, correlate. Topics include nomenclature, structure, properties, reactions, and mechanisms of. This course focuses on fundamental concepts of organic chemistry with emphasis on aliphatic hydrocarbons, alkyl halides, and alcohols. This course will enable the student to study beginning organic chemistry with emphasis However, chem*2700 and chem*3750 have been designed to provide a. Builds on concepts from ch 104 introducing basic principles of general and organic chemistry, including bonding in carbon compounds, equilibrium, stereochemistry and functional group. Organic chemistry i spring 2025 instructor: This document outlines an organic chemistry ii course that covers: Chem*2700 organic chemistry i course outline. Bonding, structure and nomenclature, stereochemistry, organic chemical reactions and mechanisms. 1) conjugated unsaturated systems including allylic compounds and dienes. However, chem*2700 and chem*3750 have been designed to provide a. Upon successful completion of chem 221, students should demonstrate an understanding of key concepts in organic chemistry including: Bonding, structure and nomenclature, stereochemistry, organic chemical reactions and mechanisms. Chm 270 or equivalent with a. Bonding, structure and nomenclature, stereochemistry, organic chemical reactions and mechanisms. Students must pass the laboratory section to receive a passing grade in the entire course. This course continues the systematic study of the theories, principles, and techniques of organic chemistry. Lewis structures and formal charge; Chm 270 or equivalent with a. This course links theoretical aspects including atomic structure, the periodic table and various forms of chemical bonding to specific classes of compounds (alkanes, alkenes, alkyl halides). This course covers bonding theories, structures,. This course continues the systematic study of the theories, principles, and techniques of organic chemistry. Adapt understanding of organic chemistry and laboratory skills to novel synthetic challenges. However,. This course is the first of a three quarter course describing the chemistry of organic (carbon containing) compounds. Lewis structures and formal charge; Bonding, structure and nomenclature, stereochemistry, organic chemical reactions and mechanisms. Bond angles and molecular shape; This course links theoretical aspects including atomic structure, the periodic table and various forms of chemical bonding to specific classes of compounds. This course will enable the student to study beginning organic chemistry with emphasis This course provides a systematic study of the theories, principles, and techniques of organic chemistry. Adapt understanding of organic chemistry and laboratory skills to novel synthetic challenges. Bonding, structure and nomenclature, stereochemistry, organic chemical reactions and mechanisms. This course links theoretical aspects including atomic structure, the periodic. This course links theoretical aspects including atomic structure, the periodic table and various forms of chemical bonding to specific classes of compounds (alkanes, alkenes, alkyl halides). This course covers bonding theories, structures,. At the end of the semester, students will be able to recognize common organic functional groups and name organic molecules using iupac nomenclature rules, correlate. Organic chemistry i. Students must pass the laboratory section to receive a passing grade in the entire course. This course is the first of a three quarter course describing the chemistry of organic (carbon containing) compounds. Chm 270 or equivalent with a. Ch 115 with a c or better. The student is expected to. This course focuses on fundamental concepts of organic chemistry with emphasis on aliphatic hydrocarbons, alkyl halides, and alcohols. This course will enable the student to study beginning organic chemistry with emphasis This course links theoretical aspects including atomic structure, the periodic table and various forms of chemical bonding to specific classes of compounds (alkanes, alkenes, alkyl halides). At the end. This course is the first of a three quarter course describing the chemistry of organic (carbon containing) compounds. This course links theoretical aspects including atomic structure, the periodic table and various forms of chemical bonding to specific classes of compounds (alkanes, alkenes, alkyl halides). Upon successful completion of chem 221, students should demonstrate an understanding of key concepts in organic. This course links theoretical aspects including atomic structure, the periodic table and various forms of chemical bonding to specific classes of compounds (alkanes, alkenes, alkyl halides). Organic chemistry i spring 2025 instructor: At the end of the semester, students will be able to recognize common organic functional groups and name organic molecules using iupac nomenclature rules, correlate. Upon successful completion. This course focuses on fundamental concepts of organic chemistry with emphasis on aliphatic hydrocarbons, alkyl halides, and alcohols. 1) conjugated unsaturated systems including allylic compounds and dienes. This course will enable the student to study beginning organic chemistry with emphasis Upon successful completion of chem 221, students should demonstrate an understanding of key concepts in organic chemistry including: Builds on concepts from ch 104 introducing basic principles of general and organic chemistry, including bonding in carbon compounds, equilibrium, stereochemistry and functional group. This course continues the systematic study of the theories, principles, and techniques of organic chemistry. This course is the first of a three quarter course describing the chemistry of organic (carbon containing) compounds. General chemistry principles ii (chm 153) or equivalent. Of key concepts in organic chemistry including: Topics include nomenclature, structure, properties, reactions, and mechanisms of. Lewis structures and formal charge; However, chem*2700 and chem*3750 have been designed to provide a. This course provides a systematic study of the theories, principles, and techniques of organic chemistry. Bilger 237a phone number (emergencies. This course covers bonding theories, structures,. Chem*2700 organic chemistry i course outline.Organic Chemistry Summary Sheet. Basics CHM215 CSU Studocu
Organic Chemistry Laboratory Course Study Material CHEM 232 Docsity
Organic Chemistry Syllabus and Course Outline
CHEM 357, Organic Chemistry, Course Outline Chem357 Studocu
Introduction to Organic Chemistry Introduction to Organic Chemistry
SCH 2121 lectiure notes UNIT CODE SCH 2108 UNIT TITLE ORGANIC
chemistry course outline part 2 Carbohydrates are one
Organic Chemistry Outline
Organic Chemistry Summary ORGANIC CHEMISTRY SUMMARY Introduction to
Chem 2122 Winter 2023 Course Outline (Long Posted) CHEM 2122
The Student Is Expected To.
Bonding, Structure And Nomenclature, Stereochemistry, Organic Chemical Reactions And Mechanisms.
At The End Of The Semester, Students Will Be Able To Recognize Common Organic Functional Groups And Name Organic Molecules Using Iupac Nomenclature Rules, Correlate.
Topics Include Nomenclature, Structure, Properties, Reactions, And Mechanisms Of.
Related Post: