Citi Refresher Course
Citi Refresher Course - Listing of rcr refresher continuing education certified modules and requirements to apply for ce credits for this course After you log into the citi site, the. Stanford provides access to required training through an interactive online tutorial, the citi. After you have completed a basic or refresher course, citi will send the vanderbilt human research protection program (vhrpp) an email within. Required tutorial on human subject research protection and good clinical practice. View details at citi program. You are required to renew your citi training every three years by taking the relevant refresher course(s). Click on the link to complete the course. Human research curriculum (satisfies irb certification requirements, not rcr) refresher course (the basic course must have been completed previously). Online rcr course through citi; Stanford provides access to required training through an interactive online tutorial, the citi. Human research curriculum (satisfies irb certification requirements, not rcr) refresher course (the basic course must have been completed previously). You are required to renew your citi training every three years by taking the relevant refresher course(s). It covers historical and current information on regulatory. Click on the link to complete the course. After you have completed a basic or refresher course, citi will send the vanderbilt human research protection program (vhrpp) an email within. It covers historical and current information. Gcp ich refresher offers retraining on gcp for clinical trials with investigational drugs and biologics (ich focus). After you log into the citi site, the. View details at citi program. View details at citi program. The refresher courses are shorter than the original training course but will contain any new or revised information a researcher would need. If you remain engaged in human subject research, the citi training refresher course must be completed every three (3) years. Gcp ich refresher offers retraining on gcp for clinical trials with investigational drugs. How do i complete the refresher course? Click on the link to complete the course. If you remain engaged in human subject research, the citi training refresher course must be completed every three (3) years. View details at citi program. Required tutorial on human subject research protection and good clinical practice. Stanford provides access to required training through an interactive online tutorial, the citi. Human research curriculum (satisfies irb certification requirements, not rcr) refresher course (the basic course must have been completed previously). Peoria irb requires its irb chairs, members, alternates and researchers to complete the citi refresher courses every three years following the initial education program. Online rcr course through. Loyola university chicago uses the online training course called the collaborative irb training initiative (citi) course to provide convenient web based training programs to the research. After you log into the citi site, the. Learners will gain a solid understanding of how to design quantitative, qualitative, and mixed methods studies, collect and analyze data, and draw meaningful conclusions across various.. Gcp ich refresher offers retraining on gcp for clinical trials with investigational drugs and biologics (ich focus). View details at citi program. You are required to renew your citi training every three years by taking the relevant refresher course(s). Loyola university chicago uses the online training course called the collaborative irb training initiative (citi) course to provide convenient web based. Loyola university chicago uses the online training course called the collaborative irb training initiative (citi) course to provide convenient web based training programs to the research. All uic investigators and key research personnel* are required to complete the initial investigator training requirements in human subject protections, a citi human subjects protections (hsp). Stanford provides access to required training through an. Online rcr course through citi; Citi program provides training courses for colleges and universities, healthcare institutions, technology and research organizations, and governmental agencies. It covers historical and current information. After you log into the citi site, the. Learners will gain a solid understanding of how to design quantitative, qualitative, and mixed methods studies, collect and analyze data, and draw meaningful. Gcp ich refresher offers retraining on gcp for clinical trials with investigational drugs and biologics (ich focus). Online rcr course through citi; If you remain engaged in human subject research, the citi training refresher course must be completed every three (3) years. It covers historical and current information. The refresher courses are shorter than the original training course but will. It covers historical and current information. The refresher courses are shorter than the original training course but will contain any new or revised information a researcher would need. After you log into the citi site, the. It covers historical and current information on regulatory. View details at citi program. Gcp ich refresher offers retraining on gcp for clinical trials with investigational drugs and biologics (ich focus). Then click the next button at the. View details at citi program. It covers historical and current information. After you log into the citi site, the. Click on the link to complete the course. It covers historical and current information on regulatory. All uic investigators and key research personnel* are required to complete the initial investigator training requirements in human subject protections, a citi human subjects protections (hsp). Then click the next button at the. Listing of rcr refresher continuing education certified modules and requirements to apply for ce credits for this course Learners will gain a solid understanding of how to design quantitative, qualitative, and mixed methods studies, collect and analyze data, and draw meaningful conclusions across various. After you have completed a basic or refresher course, citi will send the vanderbilt human research protection program (vhrpp) an email within. Citi rcr refresher stage (complete once every 2 years) will i receive a reminder email to let me know my rcr training will soon expire? The refresher courses are shorter than the original training course but will contain any new or revised information a researcher would need. Required tutorial on human subject research protection and good clinical practice. Online rcr course through citi; Citi program provides training courses for colleges and universities, healthcare institutions, technology and research organizations, and governmental agencies. You are required to renew your citi training every three years by taking the relevant refresher course(s). Human research curriculum (satisfies irb certification requirements, not rcr) refresher course (the basic course must have been completed previously). How do i complete the refresher course? It covers historical and current information.Required Ethics Training Committee on the Use of Human Subjects
CITI Social and Behavioral Research Basic.Refresher Course PDF
Training & Certification Institutional Review Board Teachers
Guide to GCP Mutual Recognition Completion Documentation
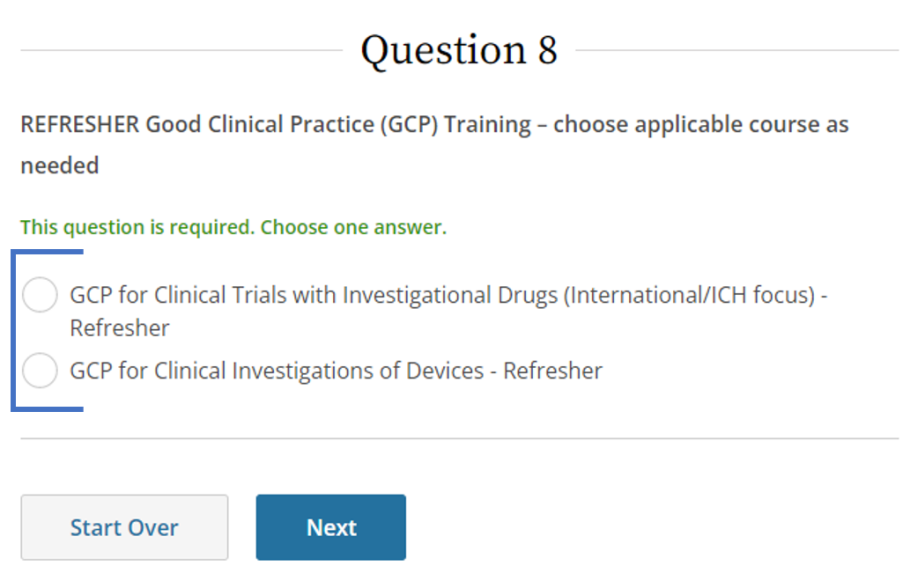
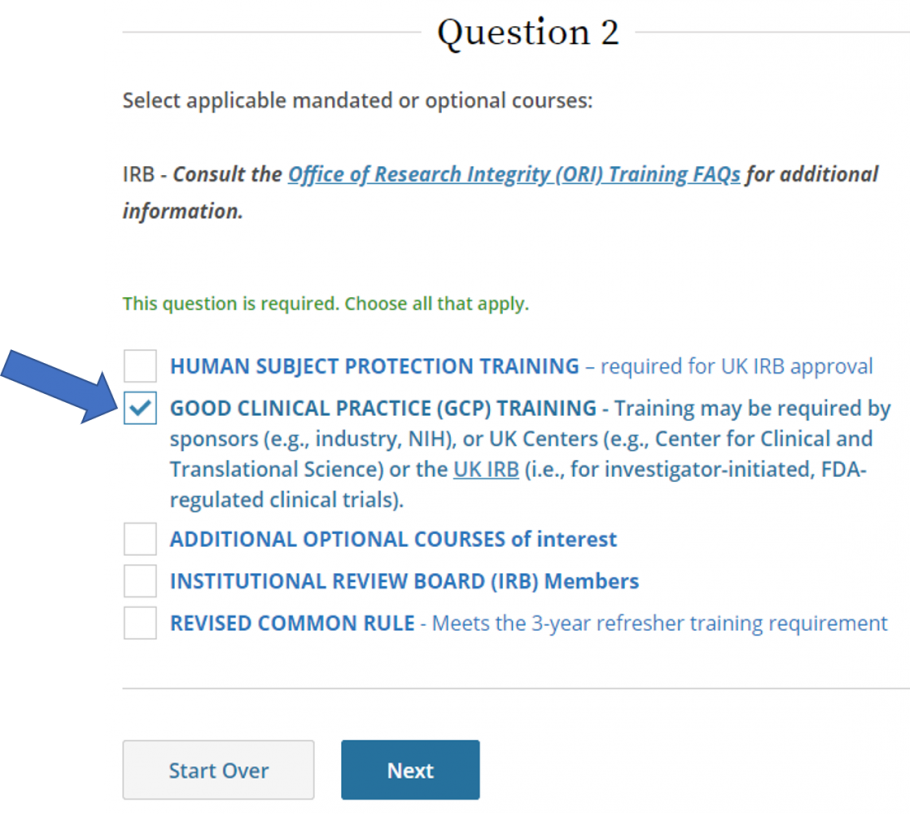
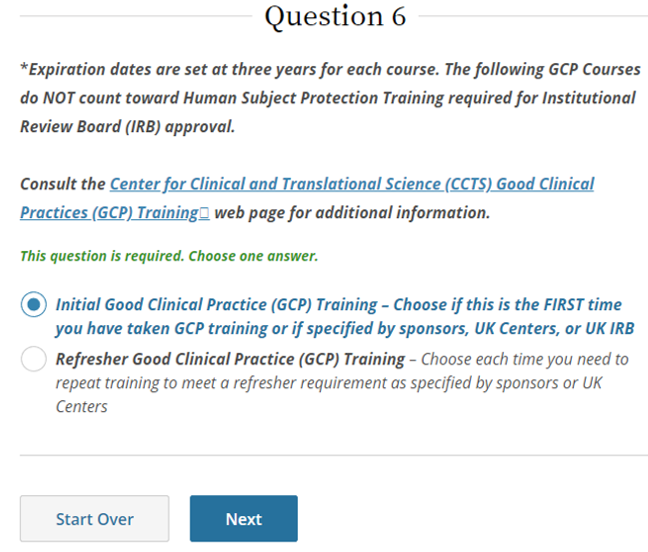
SponsorInvestigator (GCP) CITI Training FAQs University of Kentucky
SponsorInvestigator (GCP) CITI Training FAQs University of Kentucky
SponsorInvestigator (GCP) CITI Training FAQs University of Kentucky
CITI Social Behavioral Science Basic/Refresher Training Access YouTube
Reading and Understanding a CITI Program Completion Report and
Citi Training GCP and Refresher Exam Questions with Correct Answers
Stanford Provides Access To Required Training Through An Interactive Online Tutorial, The Citi.
Loyola University Chicago Uses The Online Training Course Called The Collaborative Irb Training Initiative (Citi) Course To Provide Convenient Web Based Training Programs To The Research.
It Covers Historical And Current Information.
Gcp Ich Refresher Offers Retraining On Gcp For Clinical Trials With Investigational Drugs And Biologics (Ich Focus).
Related Post: